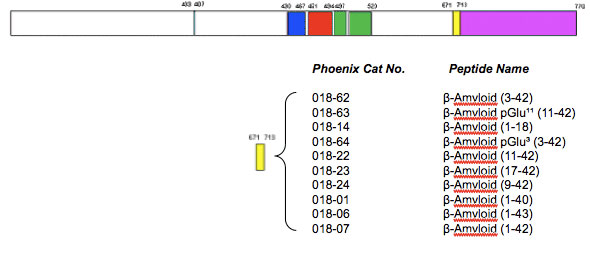
Molecular insights into the surface-catalyzed secondary nucleation of amyloid-β40 (Aβ40) by the peptide fragment Aβ16–22 | Science Advances

Stabilization of a β-hairpin in monomeric Alzheimer's amyloid-β peptide inhibits amyloid formation | PNAS

Sequence alignment of IAPP and A-(1– 40). The sequence alignment of... | Download Scientific Diagram
Aβ(1-42) tetramer and octamer structures reveal edge conductivity pores as a mechanism for membrane damage | Nature Communications

Sequence of Ab 1-42 and N-terminal truncated Ab starting at position 3.... | Download Scientific Diagram

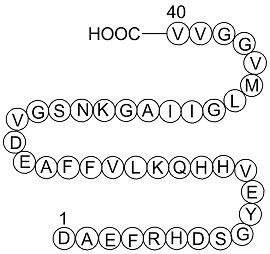
Beta-amyloid A β 1-40 amino acid sequence (active fragment A β 16-22 in... | Download Scientific Diagram

Are N- and C-terminally truncated Aβ species key pathological triggers in Alzheimer's disease? - ScienceDirect

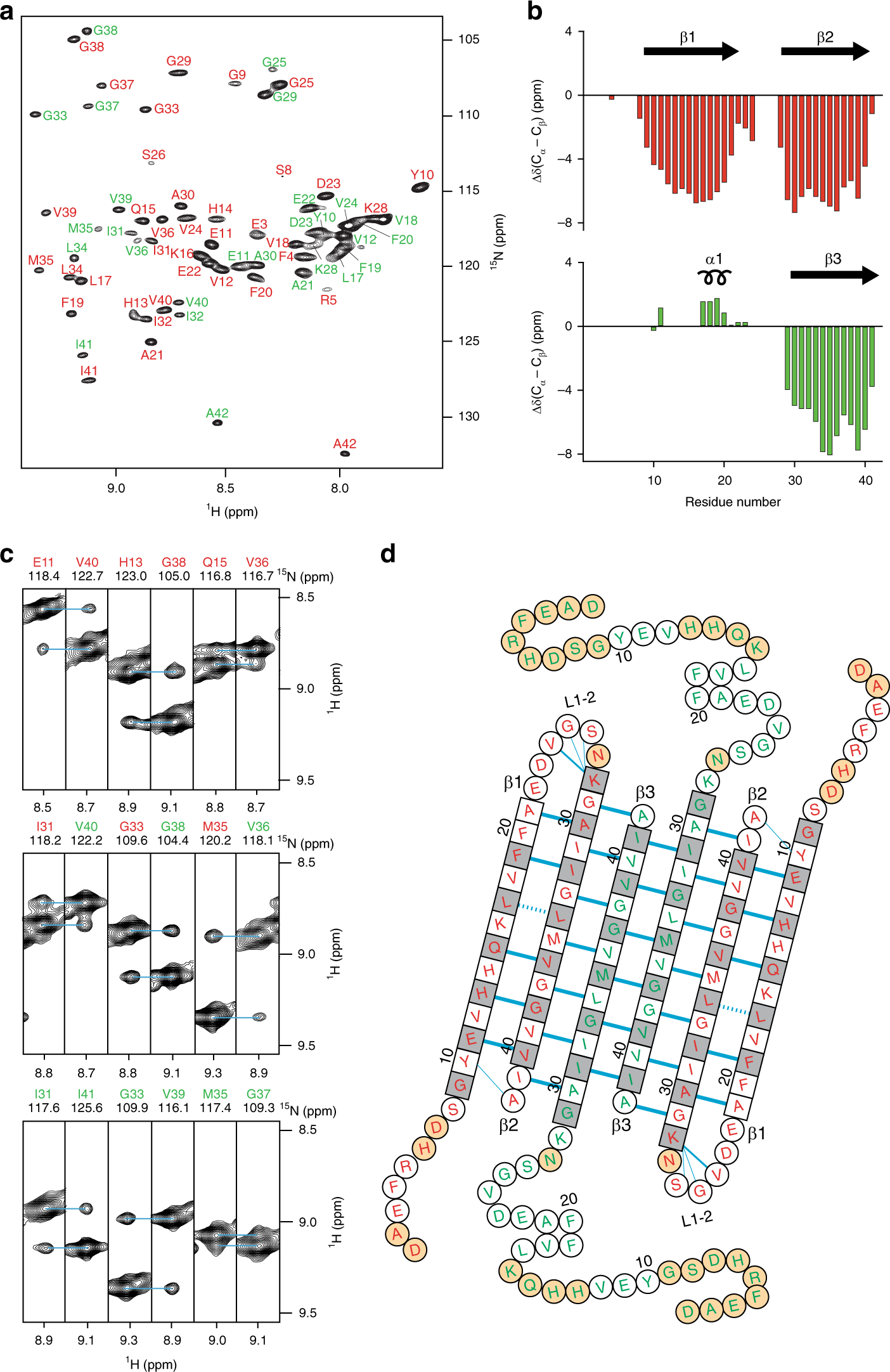
Structural conversion of neurotoxic amyloid-β1–42 oligomers to fibrils | Nature Structural & Molecular Biology

Complete aggregation pathway of amyloid β (1-40) and (1-42) resolved on an atomically clean interface | Science Advances

Dominance of Amyloid Precursor Protein Sequence over Host Cell Secretases in Determining β-Amyloid Profiles Studies of Interspecies Variation and Drug Action by Internally Standardized Immunoprecipitation/Mass Spectrometry | Journal of Pharmacology and ...
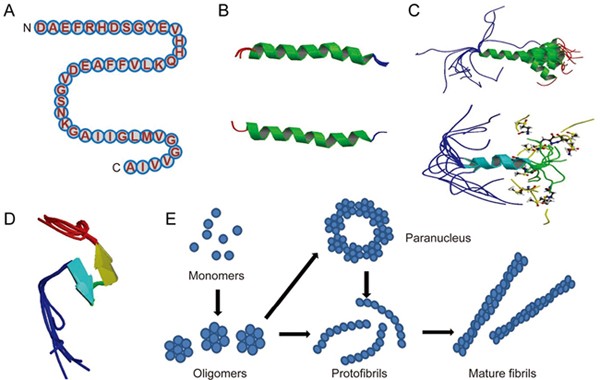
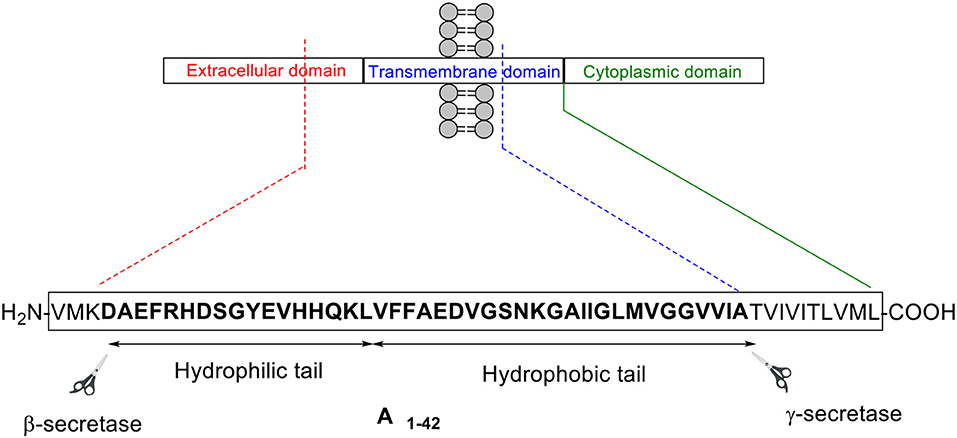
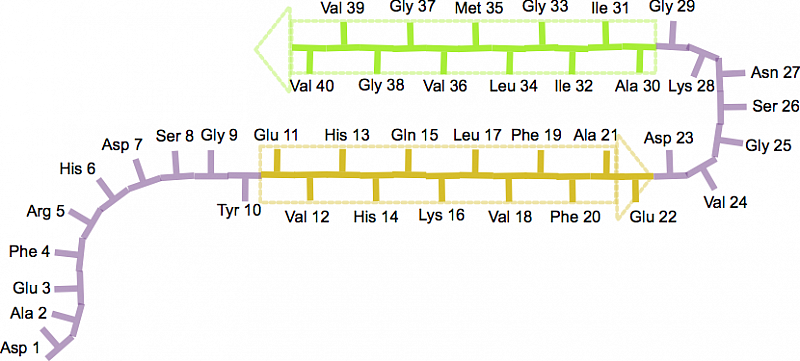
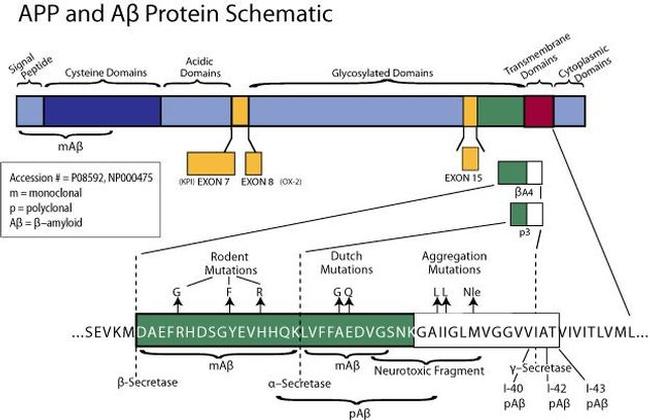
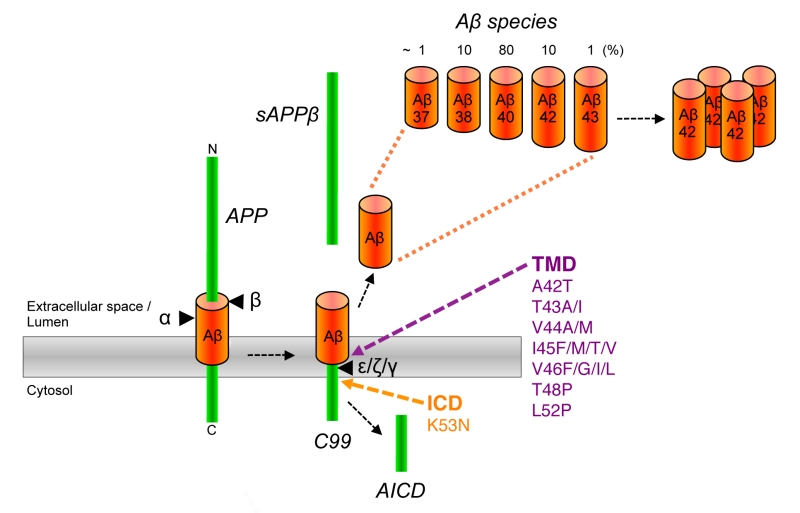
Amyloid beta: structure, biology and structure-based therapeutic development | Acta Pharmacologica Sinica

Figure 1 from How do membranes initiate Alzheimer's Disease? Formation of toxic amyloid fibrils by the amyloid β-protein on ganglioside clusters. | Semantic Scholar