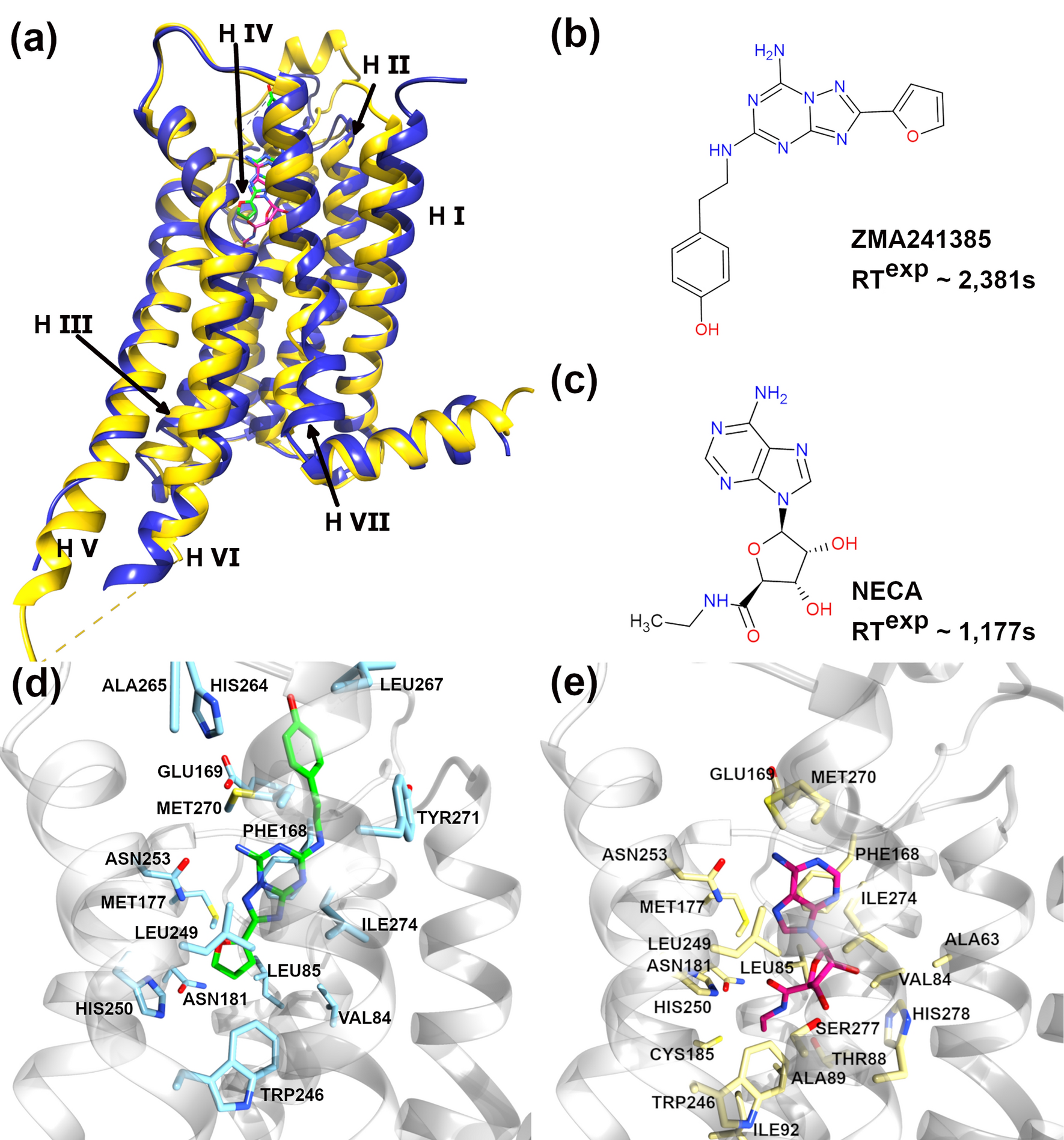
Bell-Evans model and steered molecular dynamics in uncovering the dissociation kinetics of ligands targeting G-protein-coupled receptors | Scientific Reports
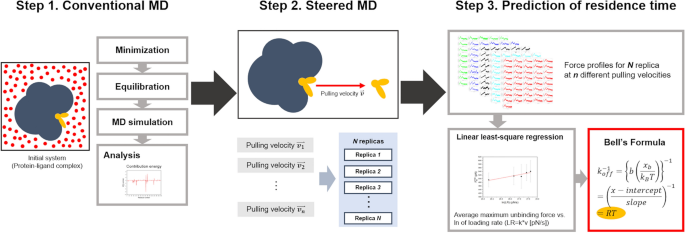
Bell-Evans model and steered molecular dynamics in uncovering the dissociation kinetics of ligands targeting G-protein-coupled receptors | Scientific Reports
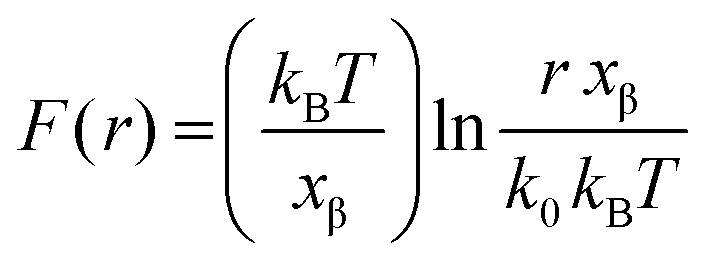
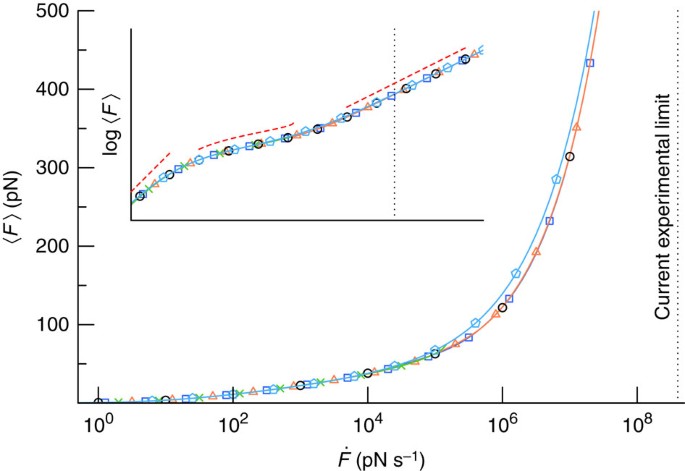
Comparison of three competing dynamic force spectroscopy models to study binding forces of amyloid-β (1–42) - Soft Matter (RSC Publishing) DOI:10.1039/C3SM52257A
Comparison of three competing dynamic force spectroscopy models to study binding forces of amyloid-β (1–42) - Soft Matter (RSC Publishing) DOI:10.1039/C3SM52257A

A) Rupture force vs. loading rate using the Bell – Evans model. Most... | Download Scientific Diagram

Bell-Evans plot of unfolding and unbinding forces. For all specific... | Download Scientific Diagram
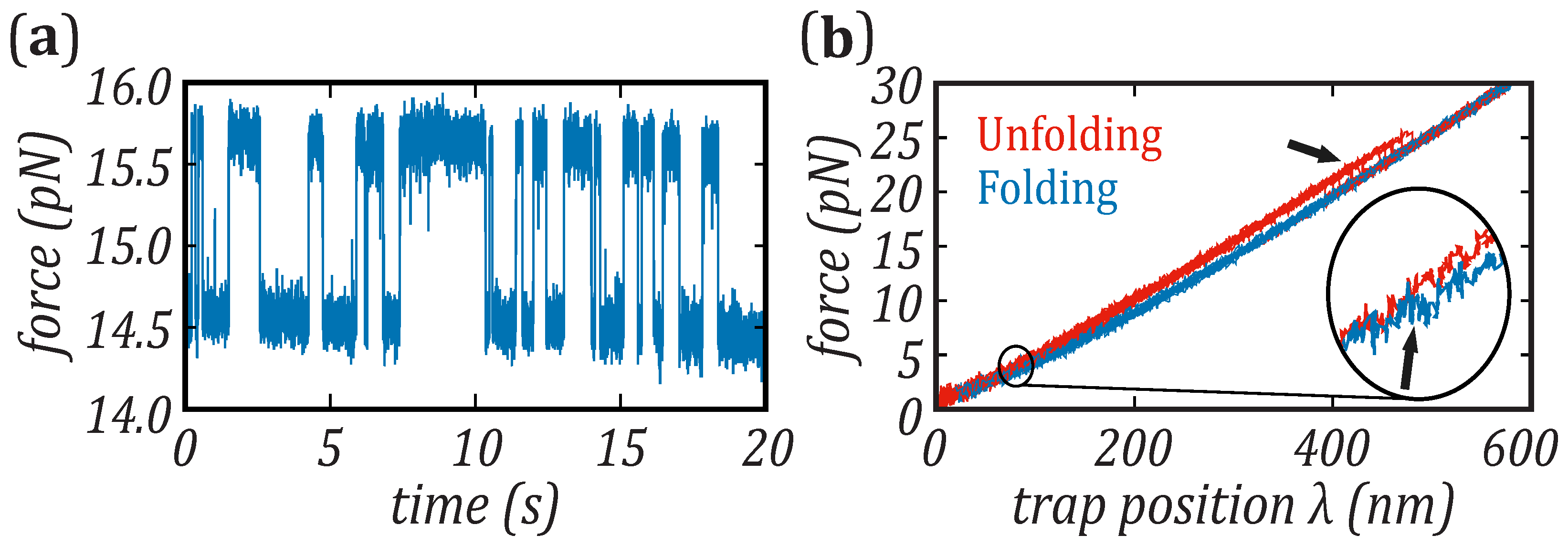
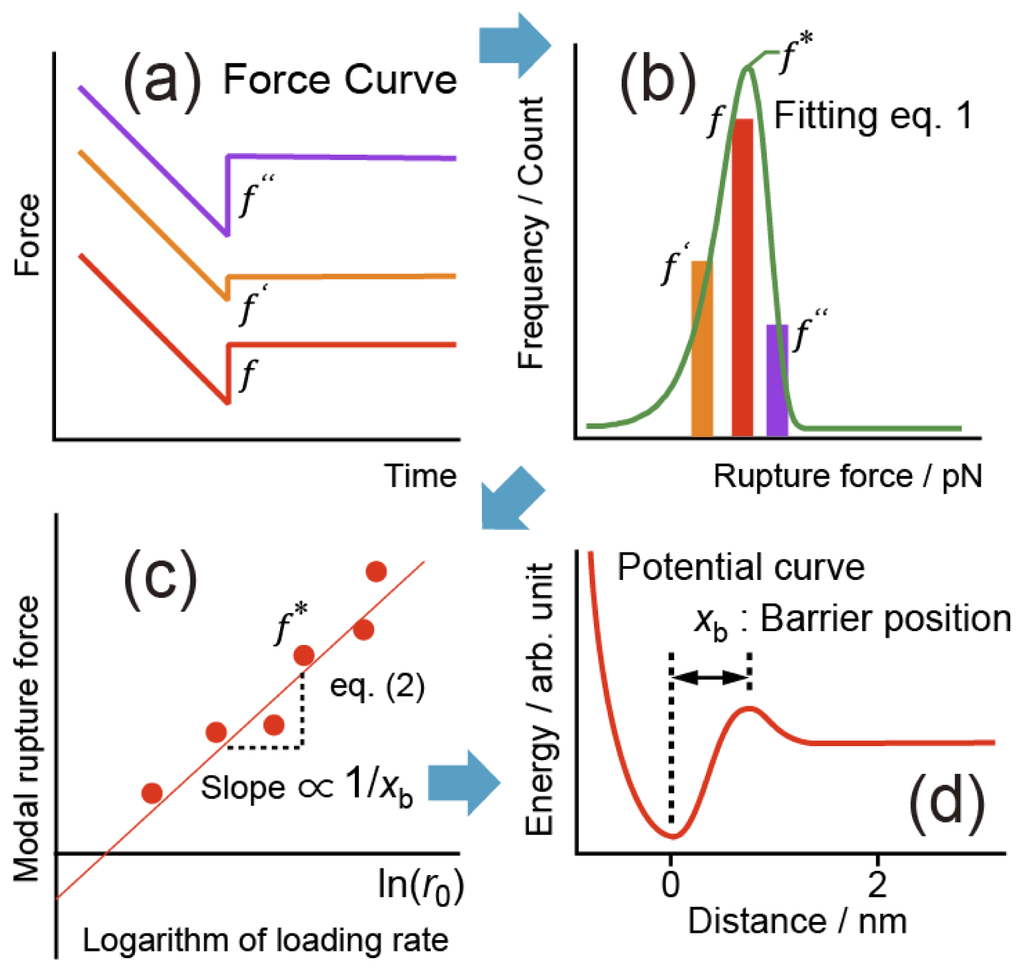
Nanomaterials | Free Full-Text | Force Dependence of Proteins' Transition State Position and the Bell–Evans Model
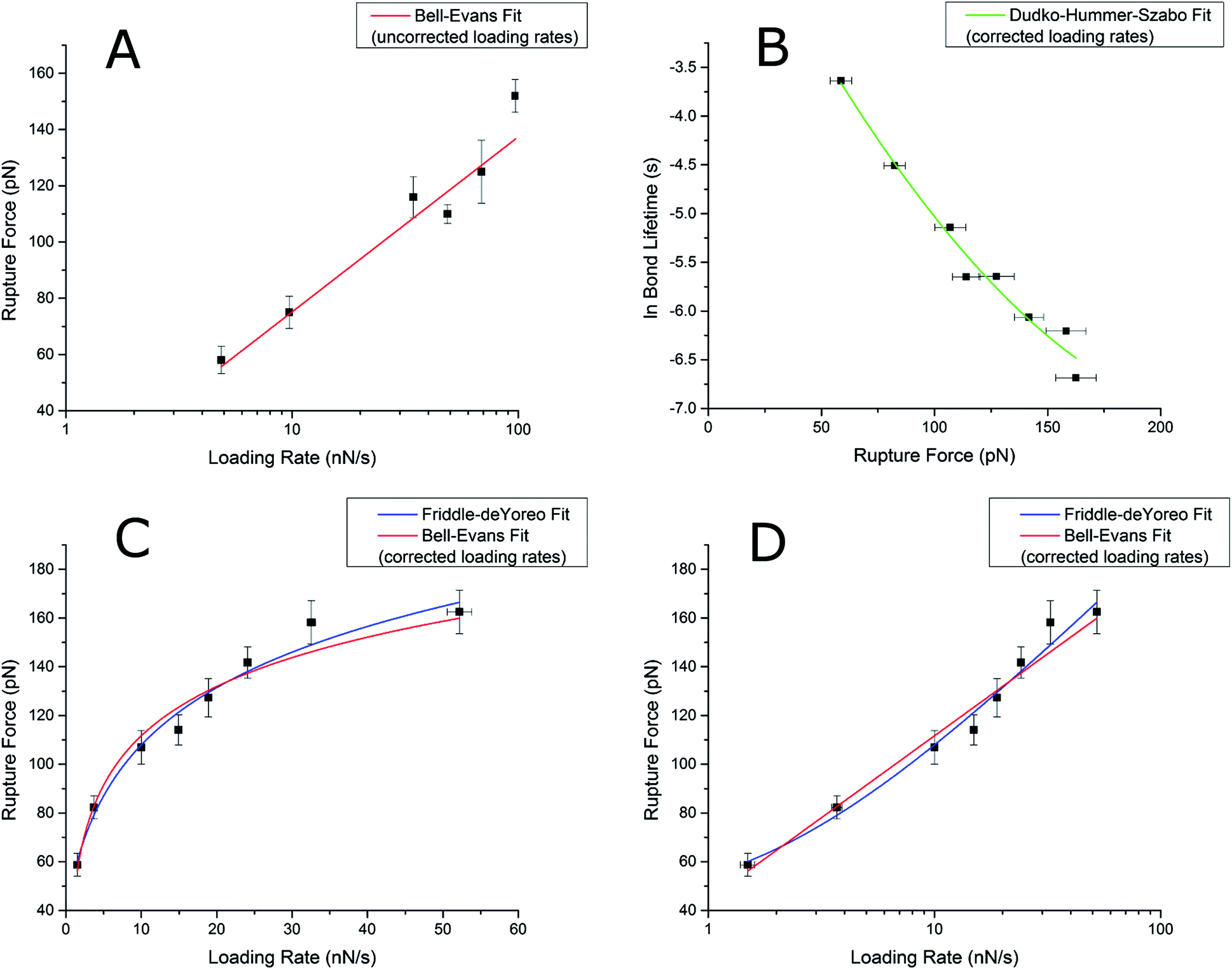
Comparison of three competing dynamic force spectroscopy models to study binding forces of amyloid-β (1–42) - Soft Matter (RSC Publishing) DOI:10.1039/C3SM52257A

Nanomaterials | Free Full-Text | Force Dependence of Proteins' Transition State Position and the Bell–Evans Model
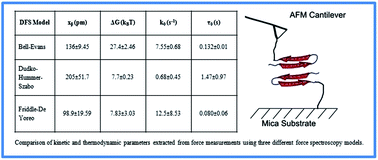
Comparison of three competing dynamic force spectroscopy models to study binding forces of amyloid-β (1–42) - Soft Matter (RSC Publishing)

Extending Bell's Model: How Force Transducer Stiffness Alters Measured Unbinding Forces and Kinetics of Molecular Complexes - ScienceDirect
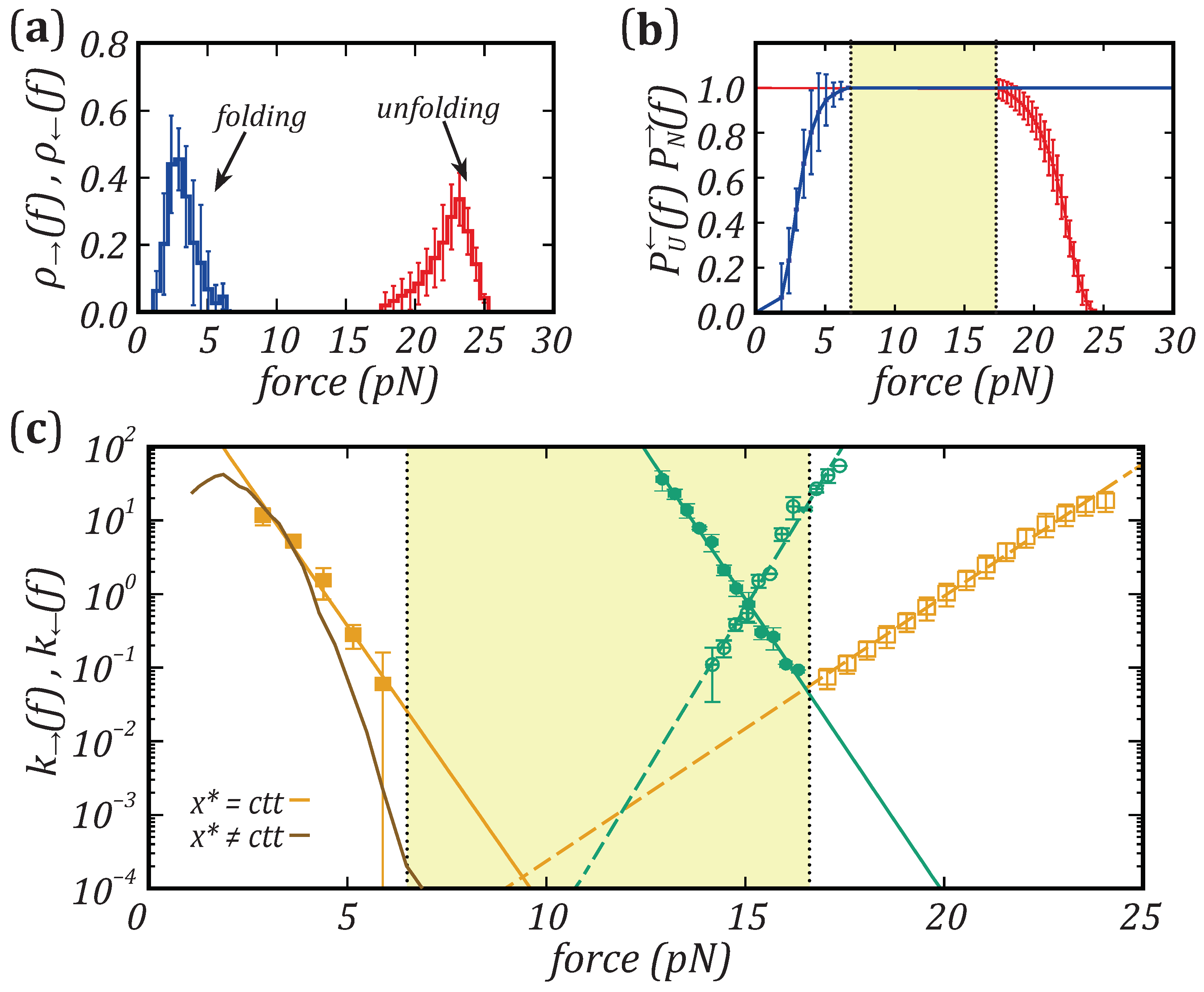
Nanomaterials | Free Full-Text | Force Dependence of Proteins' Transition State Position and the Bell–Evans Model

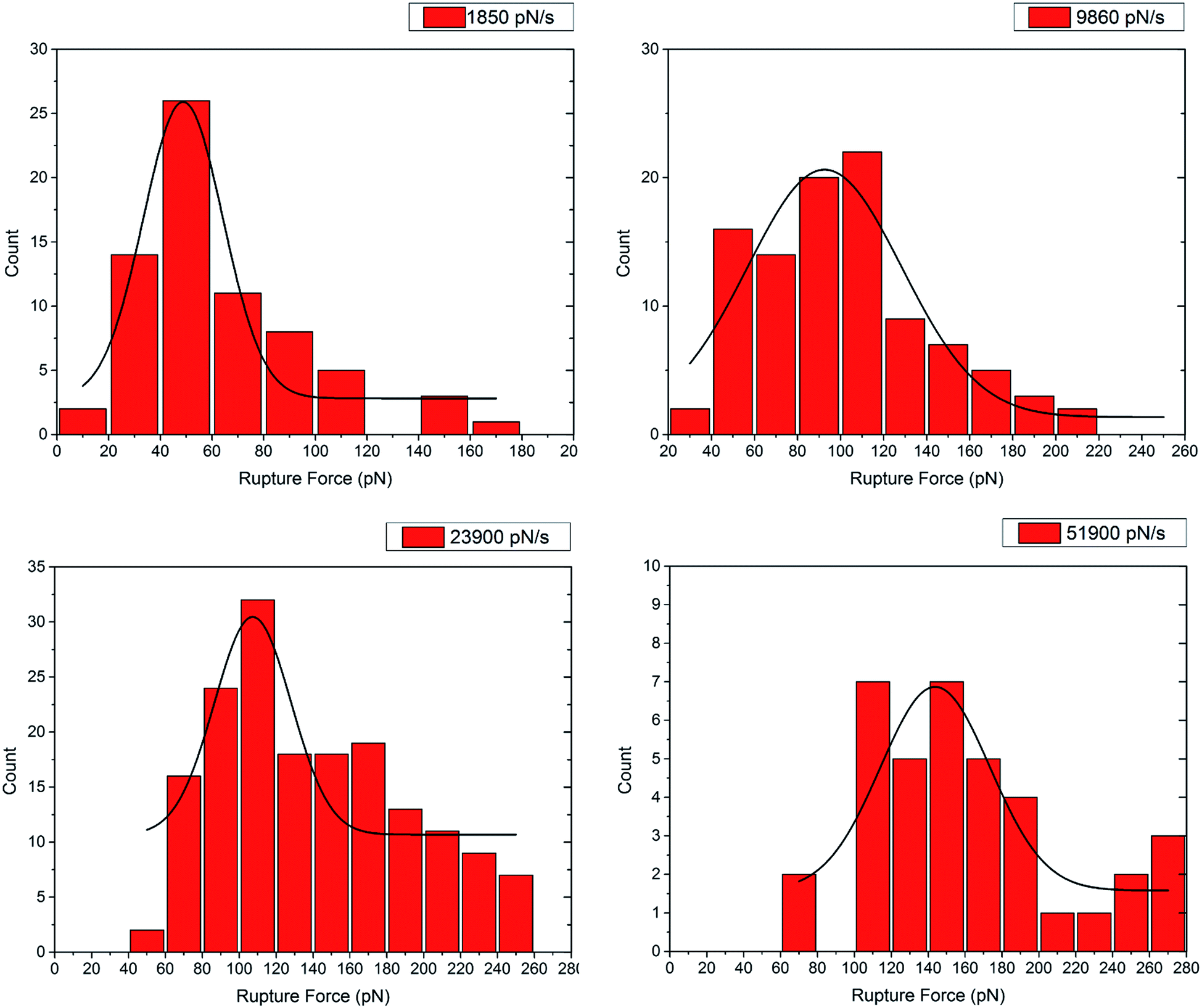
Dependence of the most probable and average bond rupture force on the force loading rate: First order correction to the Bell–Evans model: Journal of Applied Physics: Vol 114, No 3

Bell-Evans plot showing a linear relationship between the most probable... | Download Scientific Diagram