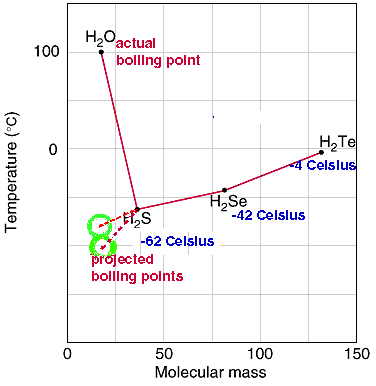
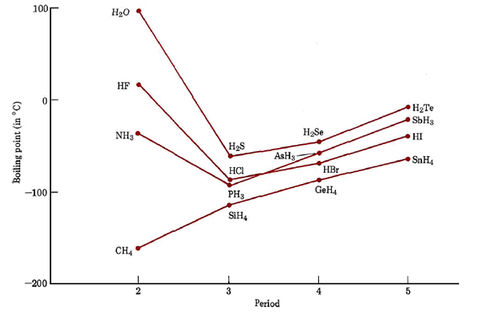
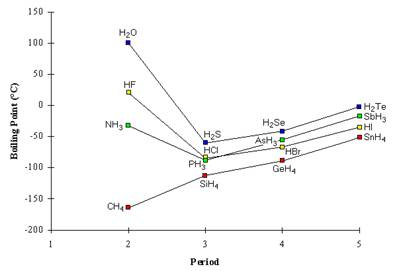
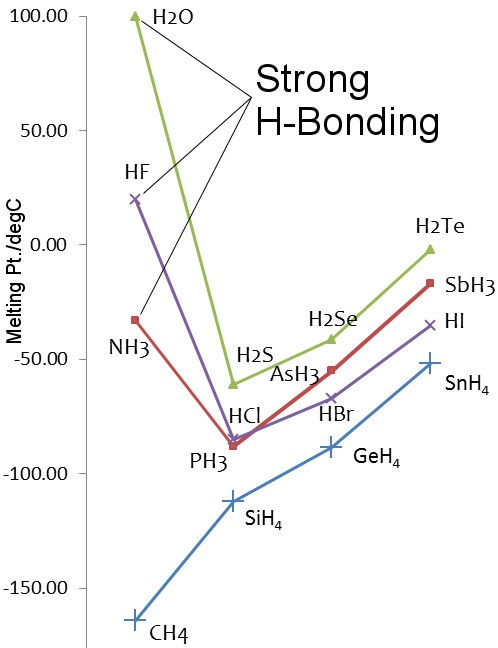
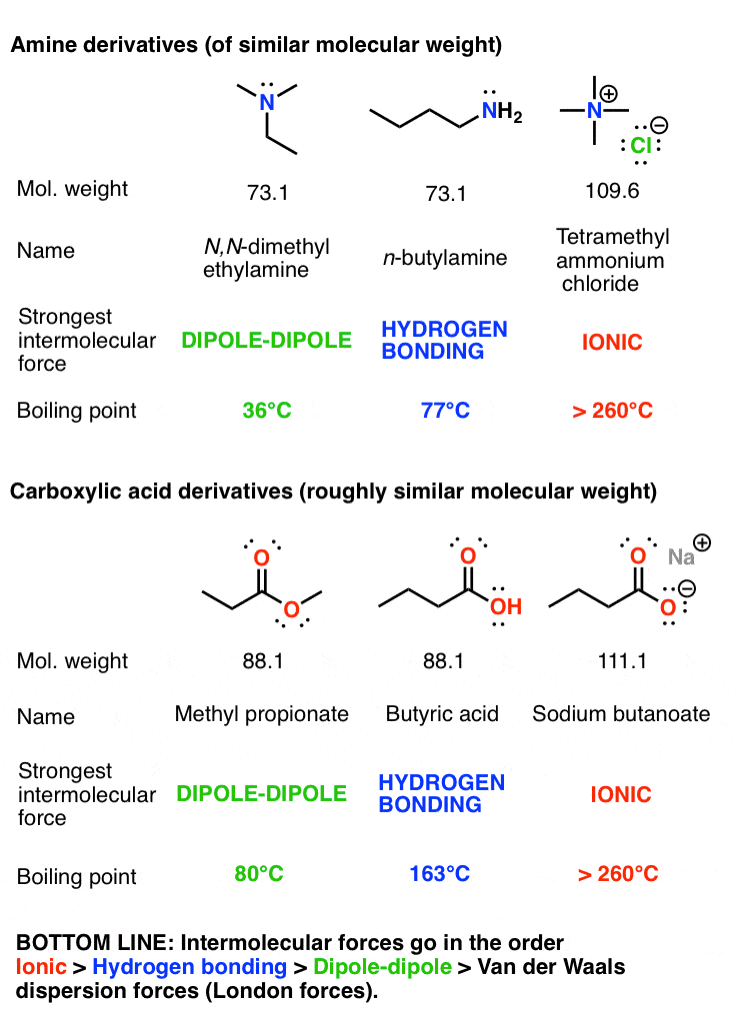
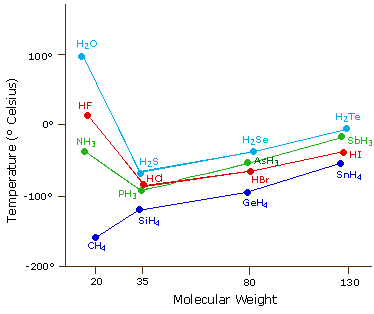
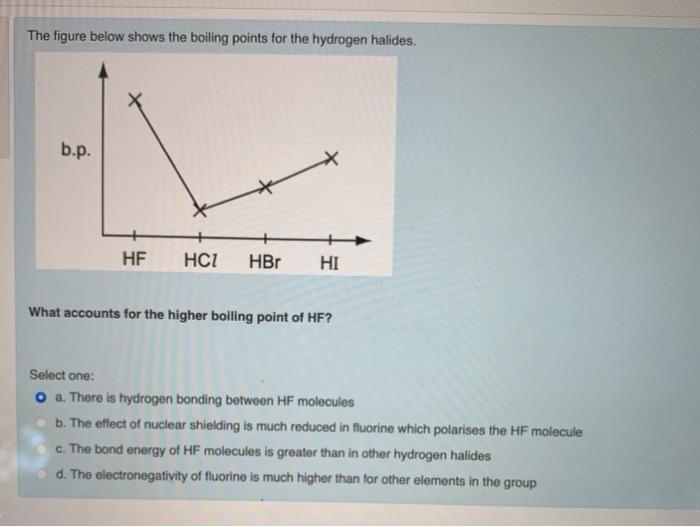
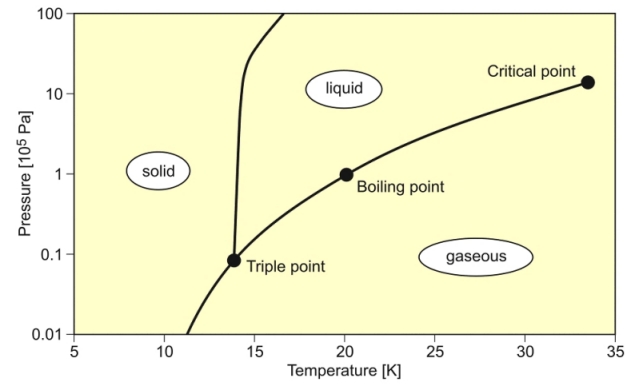
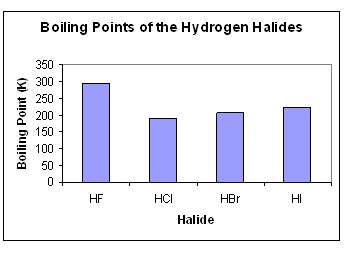
SOLVED: Q6: Unit 10 Level 2 The boiling point of water is about 2008C higher than one would predict from the boiling points of hydrogen sulfide and hydrogen selenide. One may explain

Full Frame Macro Close Up Photo of Oxygen and Hydrogen Electrolysis and Boiling Point. Stock Photo - Image of effervescence, liquid: 176172552
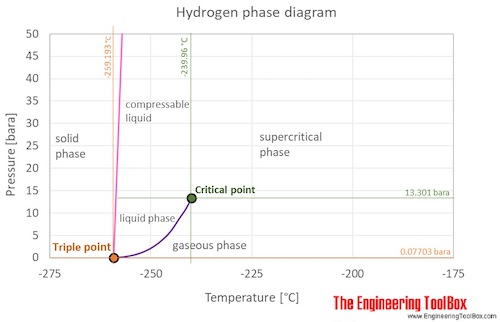
thermodynamics - Boiling point of hydrogen: relation between pressure and temperature near of triple point - Physics Stack Exchange

Elements Hydrogen Number of: Protons 1 Neutrons 0 Electrons 1 Boiling point -252 Freezing point -259 State at room temperature gas. - ppt download