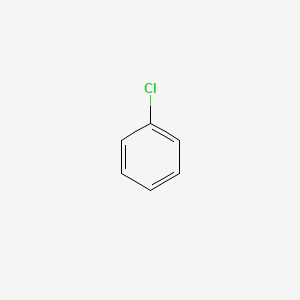
Difference Between Chlorobenzene and Cyclohexyl Chloride | Compare the Difference Between Similar Terms
1. A mixture is chlorobenzene and water (which are virtually immiscible) boils at 90.3^° C at an external pressure of 740.2mm. The vapour pressure of pure water at 40.3^° C if 530.1mm.

Correct order of boiling point for the given compounds is:a) 1 - chloropropaneb) isopropyl chloridec) 1 - chlorobutane

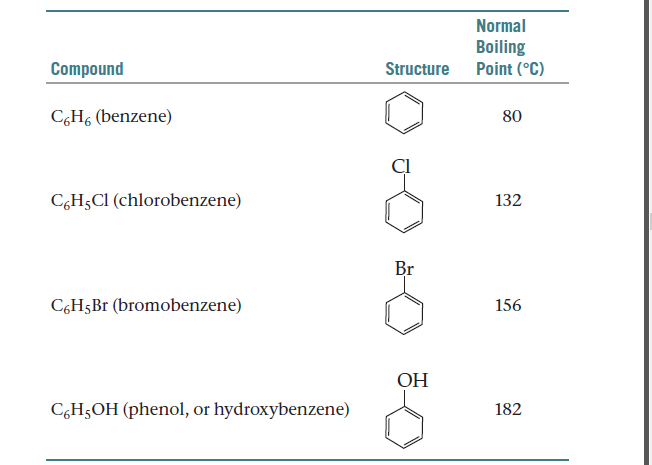
Arrange each set of compounds in the order increasing boiling points. (I) bromobenzene, chlorobenzene, - Brainly.in
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

Difference Between Chlorobenzene and Benzyl Chloride | Definition, Chemical Properties, Uses, Differences
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

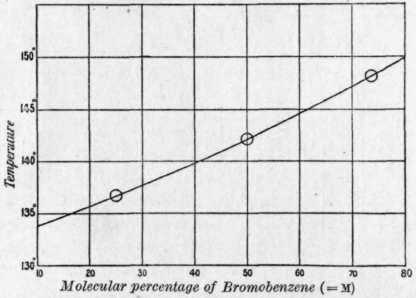
Boiling point T b ,°С, vs. molecular weight M: (1): (a) benzene, (b)... | Download Scientific Diagram
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

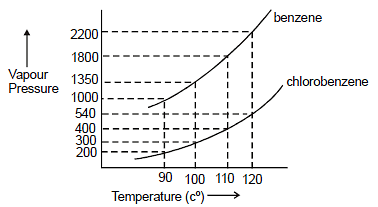
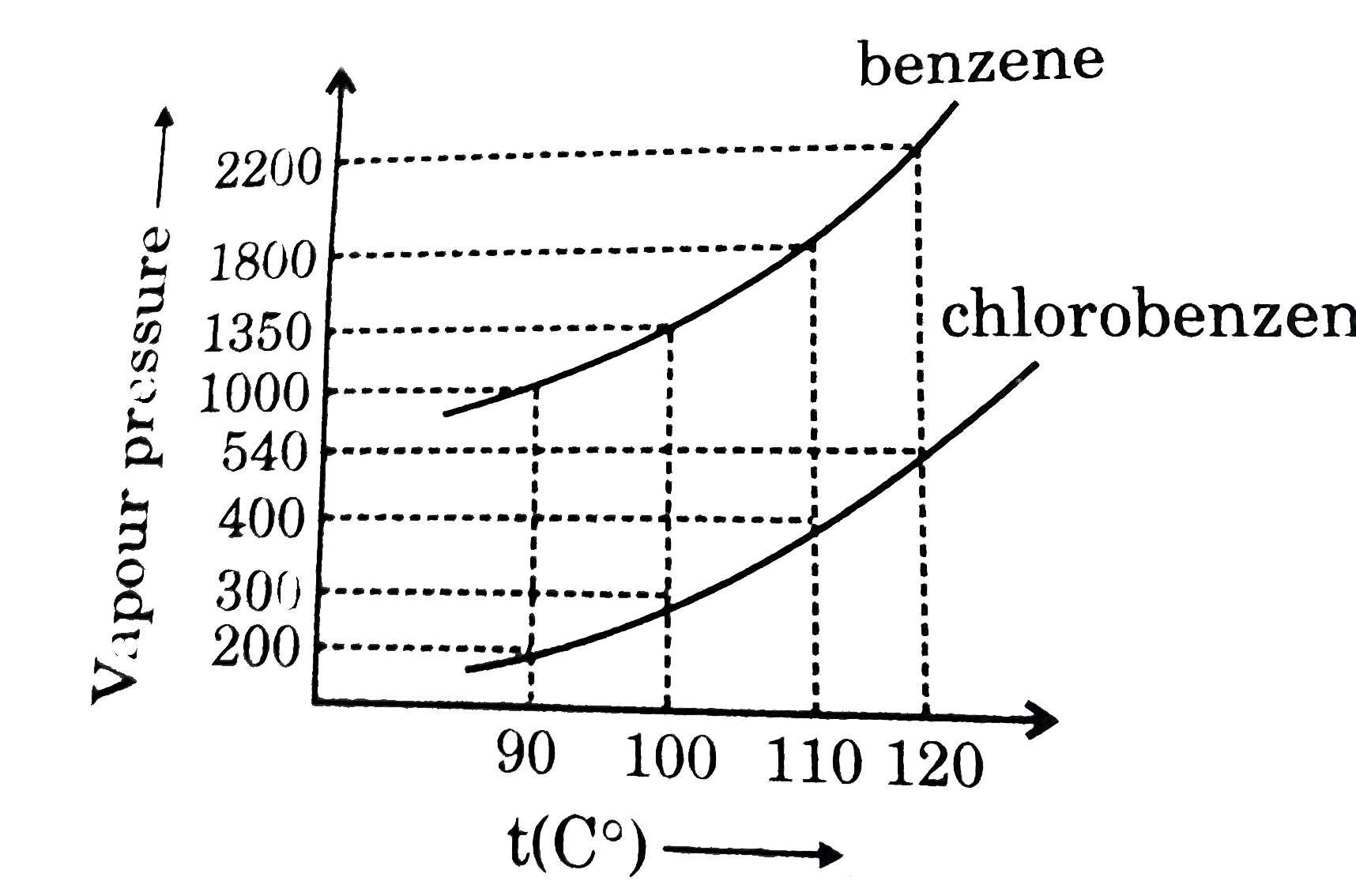
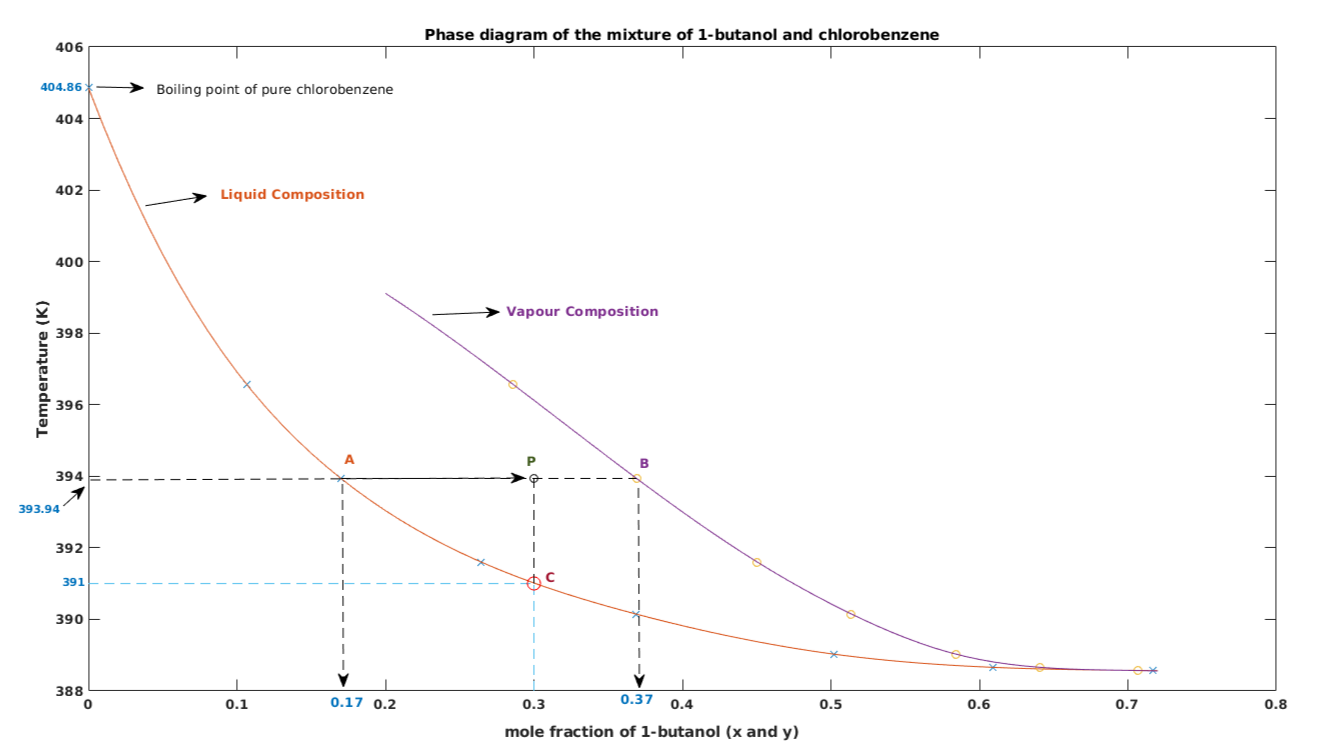
Assuming the formation of an ideal solution, the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar' mass = 112.5)' using the given

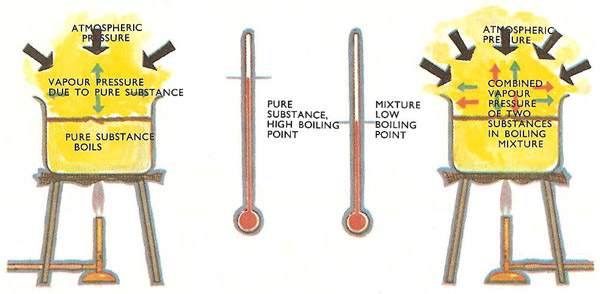
SOLVED: ii A mixture of liquid chlorobenzene and water is boiling at 130 mm Hg pressure. Liquid chlorobenzene is immiscible in water.Determine the boiling point of the mixture in K and the