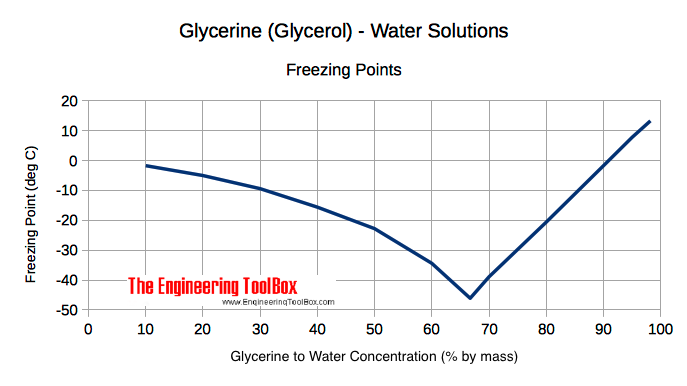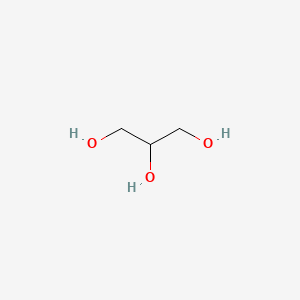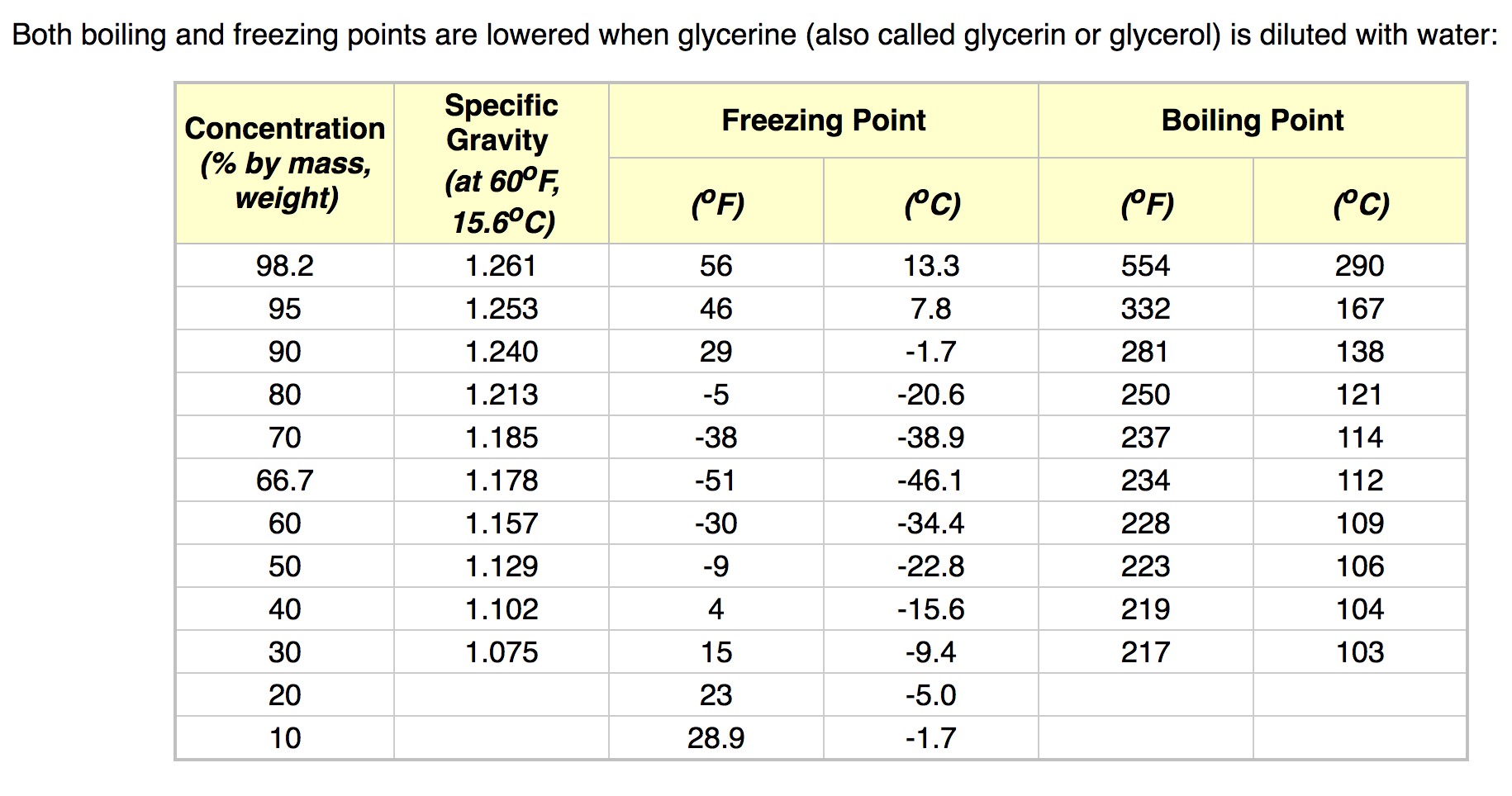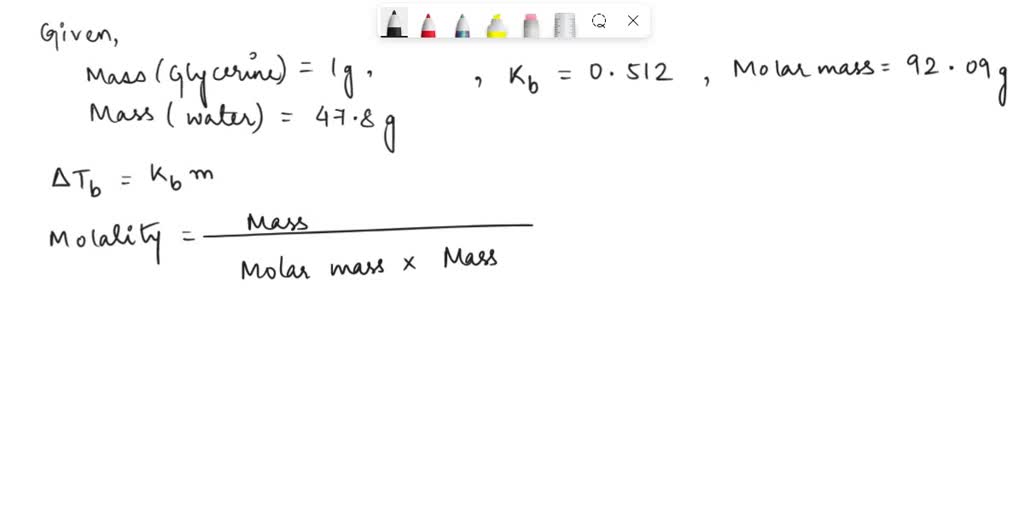
SOLVED: What is the boiling point of a solution of 1.0 g of glycerin, C3H5(OH)3, in 47.8 g of water? Assume an ideal solution.

Boiling points of the propylene glycol + glycerol system at 1 atmosphere pressure: 188.6-292 °C without and with added water or nicotine. - Abstract - Europe PMC