EP0183110B1 - Azeotrope-like compositions of trichlorotrifluoroethane, ethanol, acetone, nitromethane and hexane - Google Patents

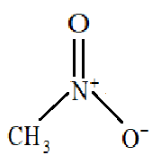
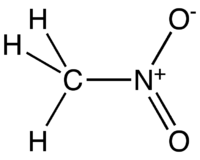
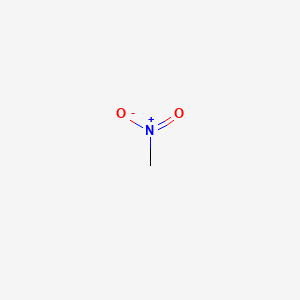
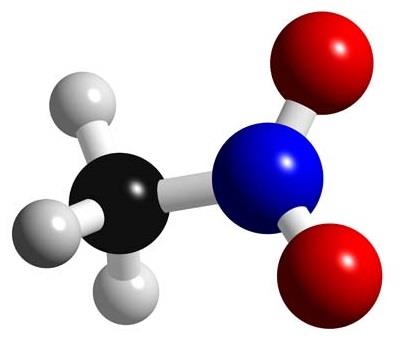
Nitromethane has a relatively high boiling point of around 100 degrees C. Which of the following intermolecular force is not present in pure liquid nitromethane and why? A) Van der Waals force.
Promotion effect of nitromethane on ethanol ignition as a function of... | Download Scientific Diagram

PDF) Boiling point estimation of 1-butanol and nitromethane by the viscosity- temperature dependence study of corresponding binary fluid systems