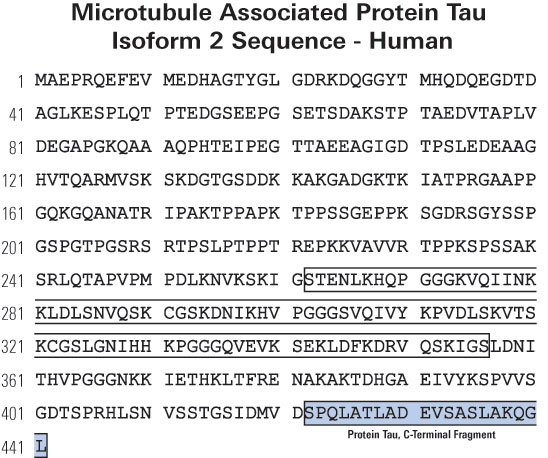
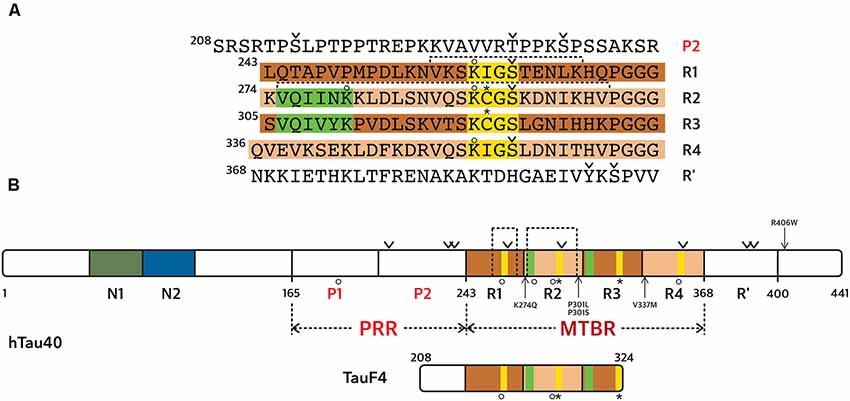
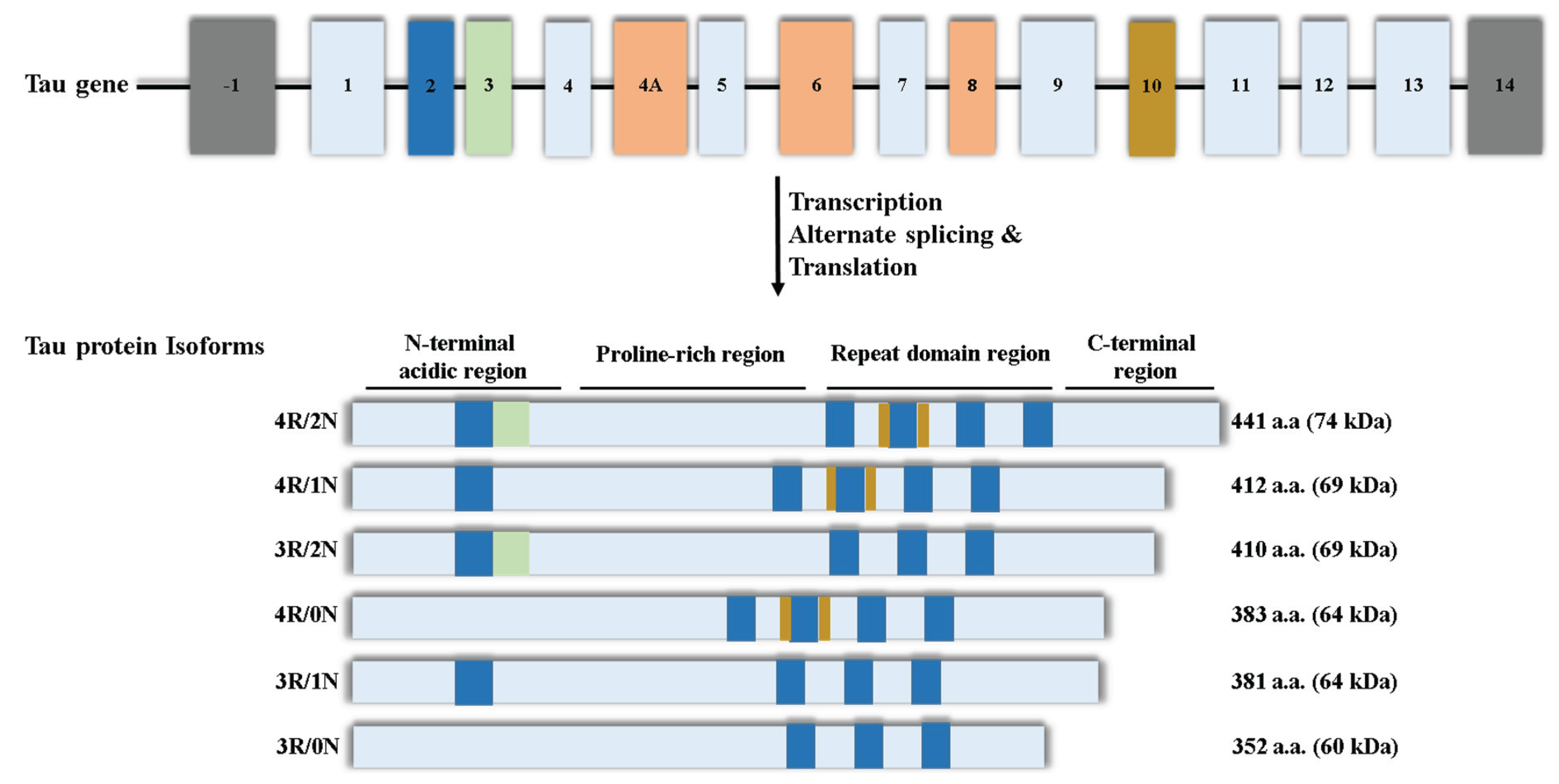
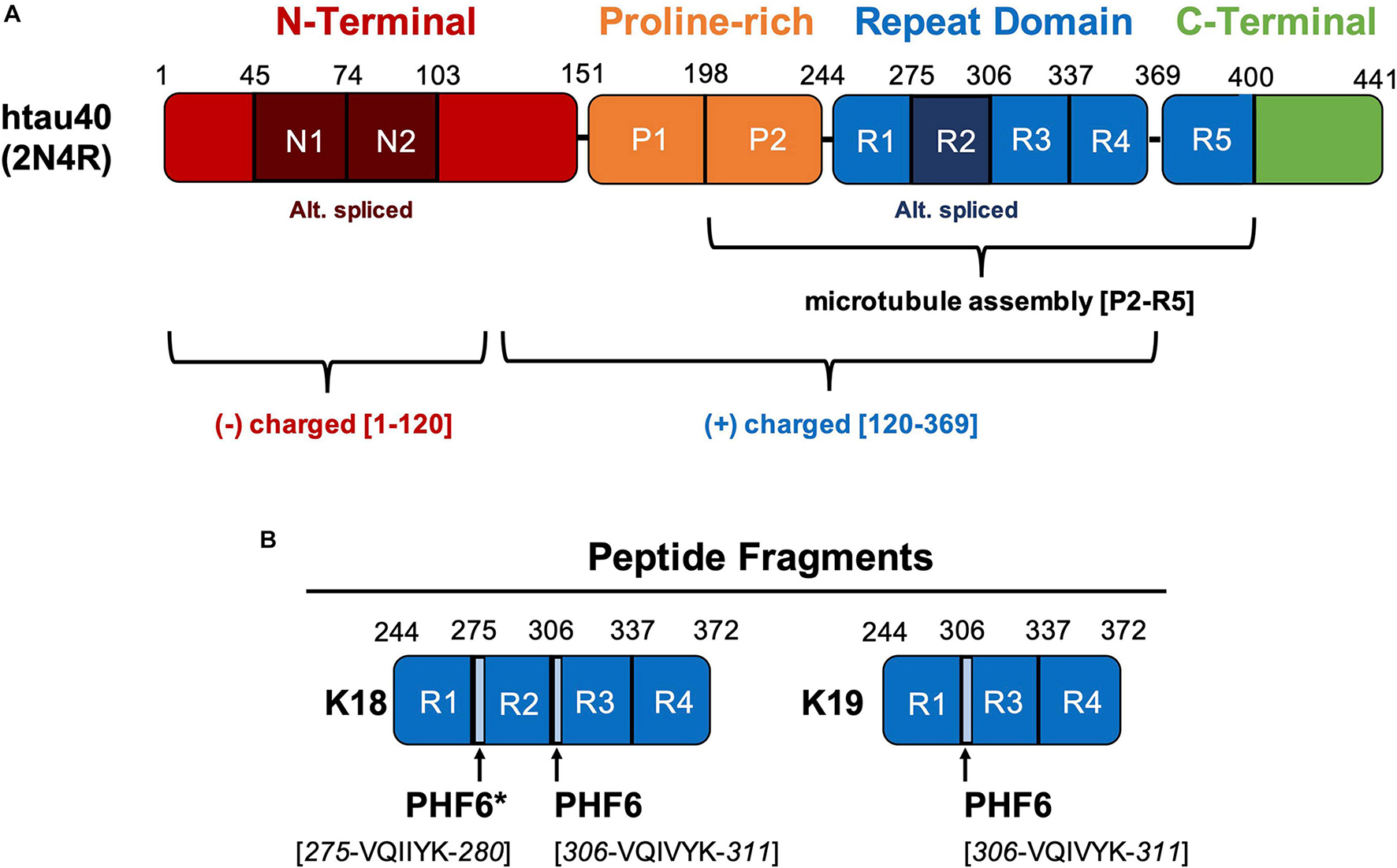
We reported the full-length amino acid sequence of the human and mouse... | Download Scientific Diagram

Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer's continuum when only subtle changes in Aβ pathology are detected | EMBO Molecular Medicine
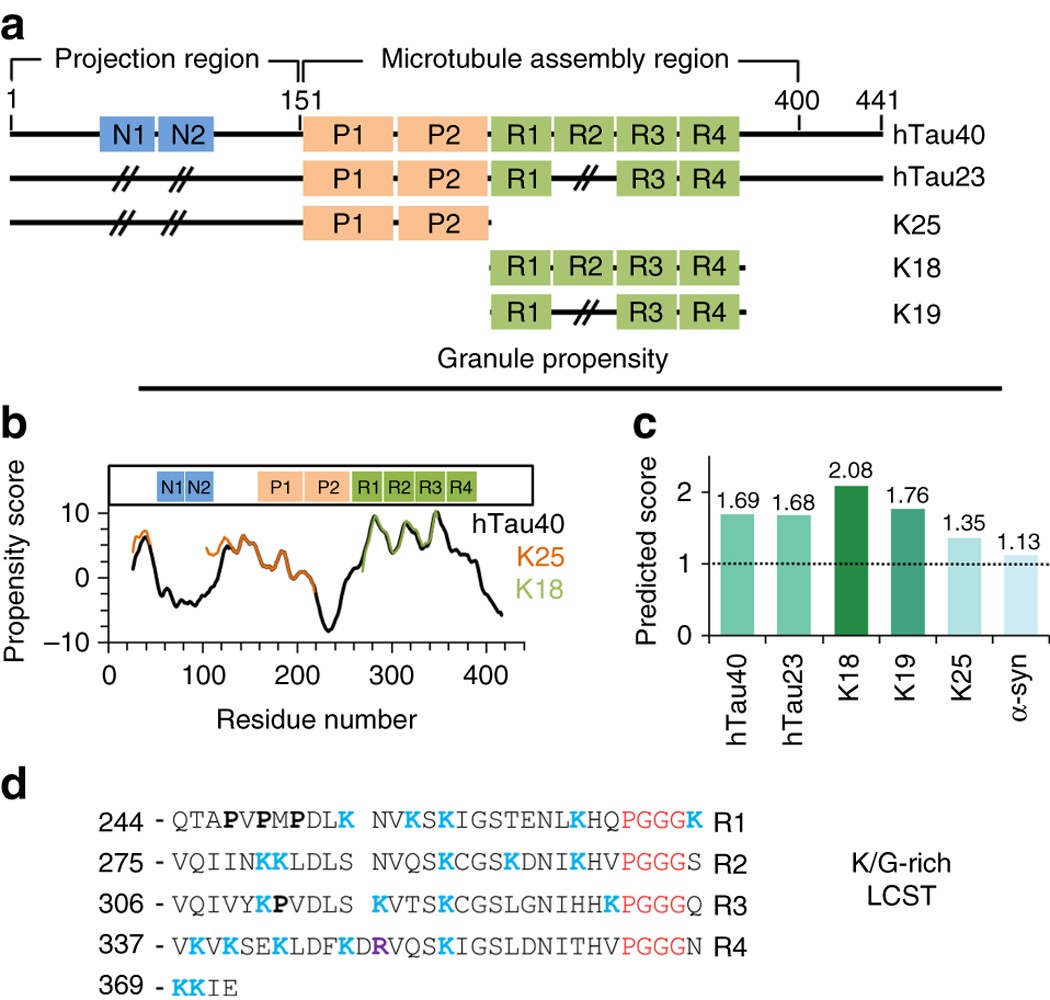
Liquid–liquid phase separation of the microtubule-binding repeats of the Alzheimer-related protein Tau | Nature Communications
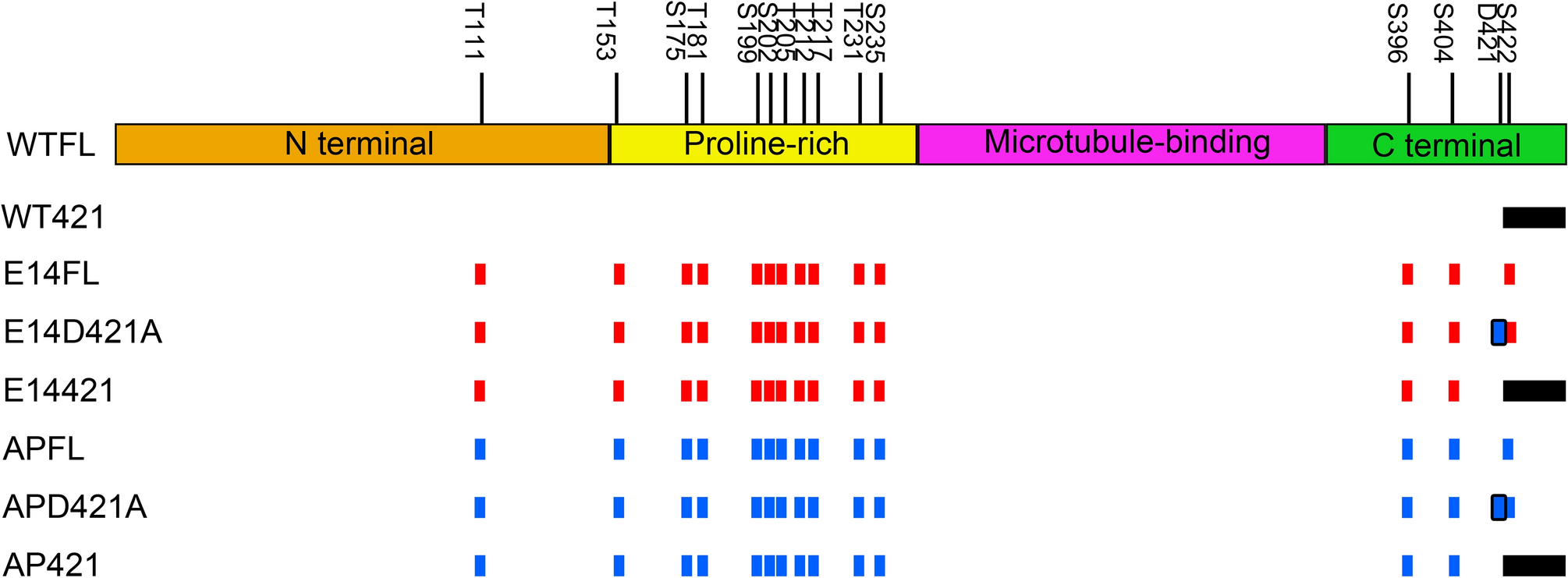
Cleavage of human tau at Asp421 inhibits hyperphosphorylated tau induced pathology in a Drosophila model | Scientific Reports
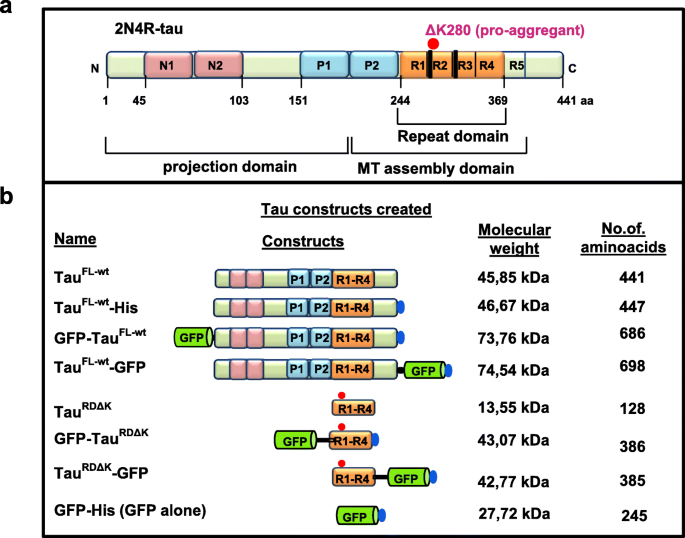
FRET-based Tau seeding assay does not represent prion-like templated assembly of Tau filaments | Molecular Neurodegeneration | Full Text

Phosphorylation of Tau Protein Associated as a Protective Mechanism in the Presence of Toxic, C-Terminally Truncated Tau in Alzheimer's Disease | IntechOpen

Heterotypic electrostatic interactions control complex phase separation of tau and prion into multiphasic condensates and co-aggregates | PNAS
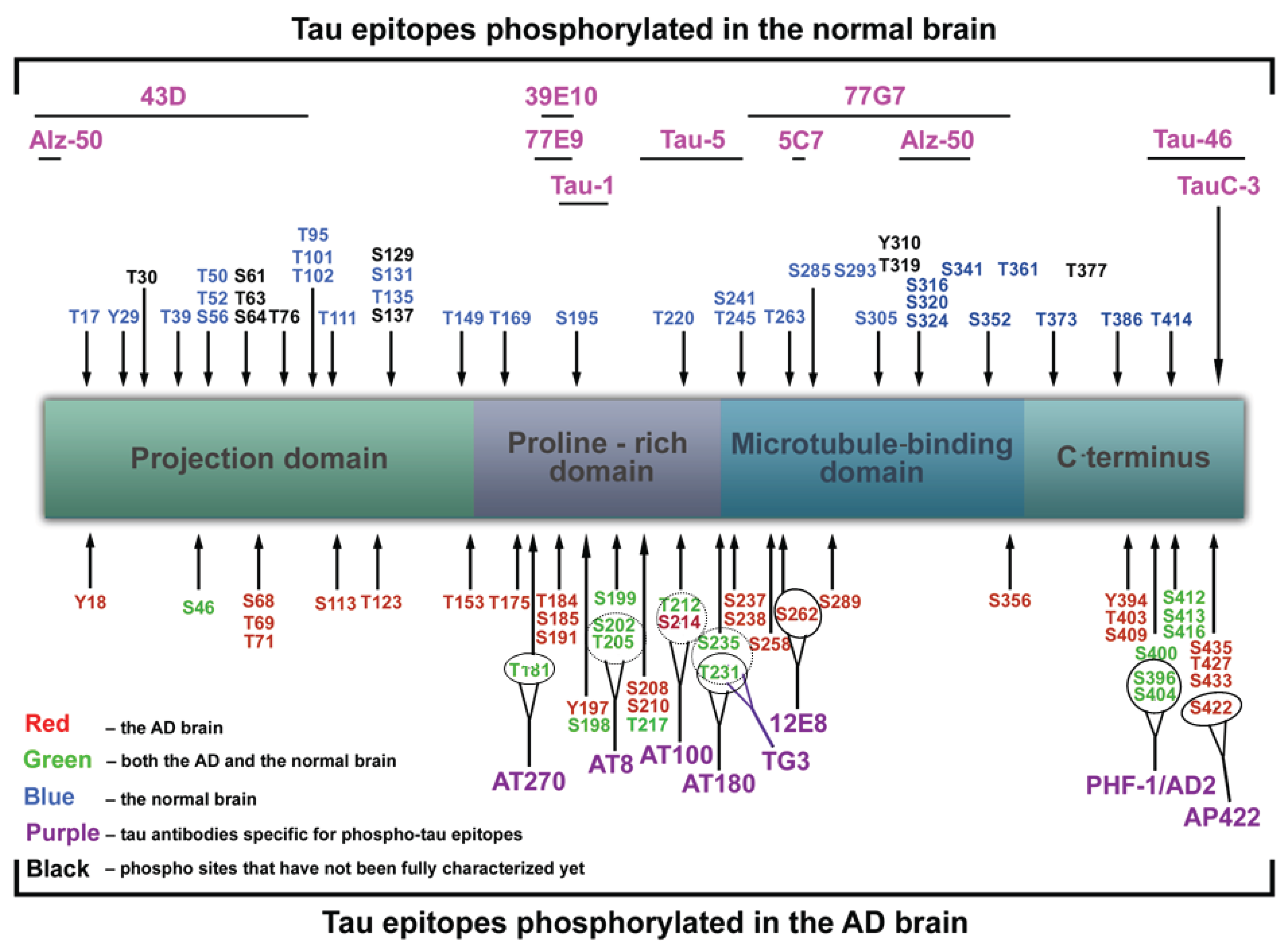
Biomolecules | Free Full-Text | Tau Protein Hyperphosphorylation and Aggregation in Alzheimer's Disease and Other Tauopathies, and Possible Neuroprotective Strategies

Amino acid sequence of the longest tau isoform (441 amino acids). N1... | Download Scientific Diagram

Tau Protein Quantification in Human Cerebrospinal Fluid by Targeted Mass Spectrometry at High Sequence Coverage Provides Insights into Its Primary Structure Heterogeneity | Journal of Proteome Research

Tau protein sequence and domain organization. The sequence numbering... | Download Scientific Diagram

Biochemical classification of tauopathies by immunoblot, protein sequence and mass spectrometric analyses of sarkosyl-insoluble and trypsin-resistant tau | SpringerLink