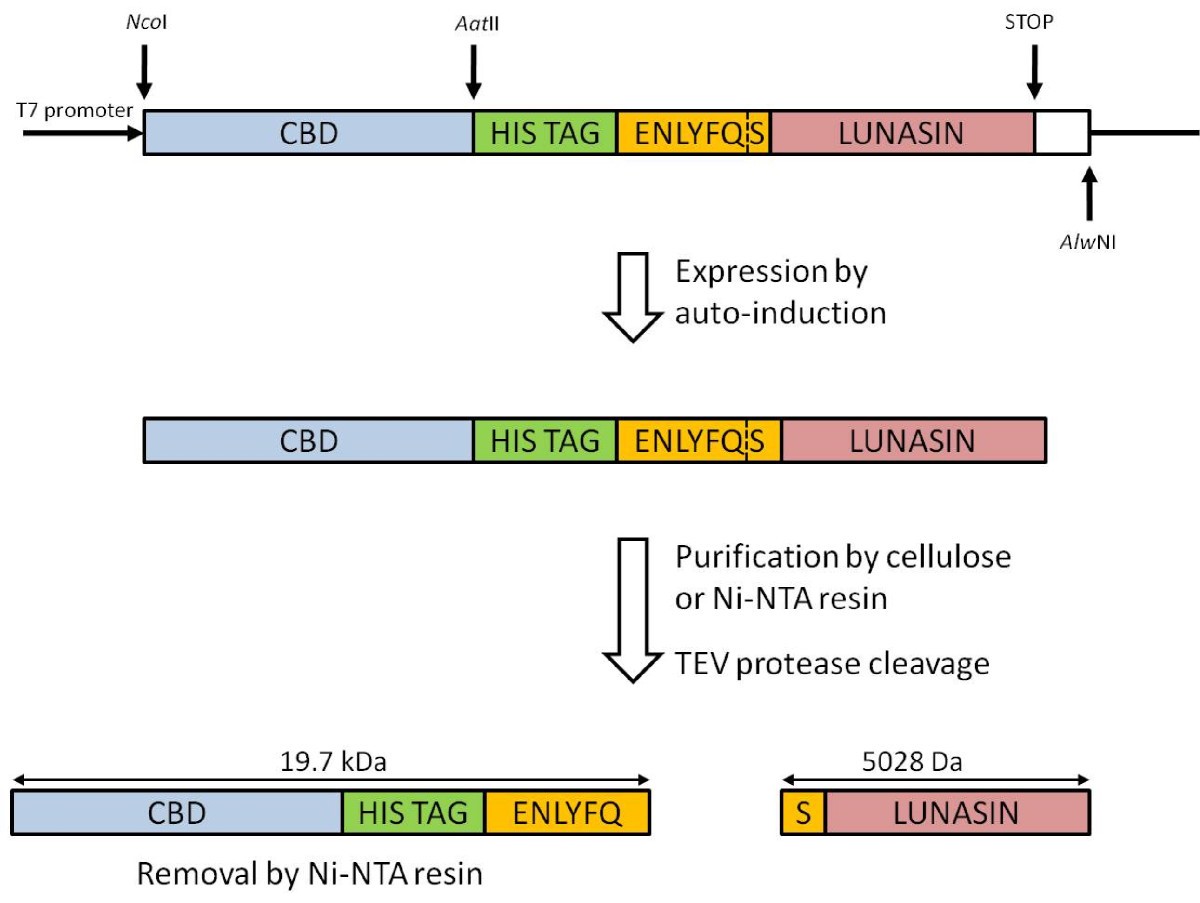
Highly efficient soluble expression, purification and characterization of recombinant Aβ42 from Escherichia coli
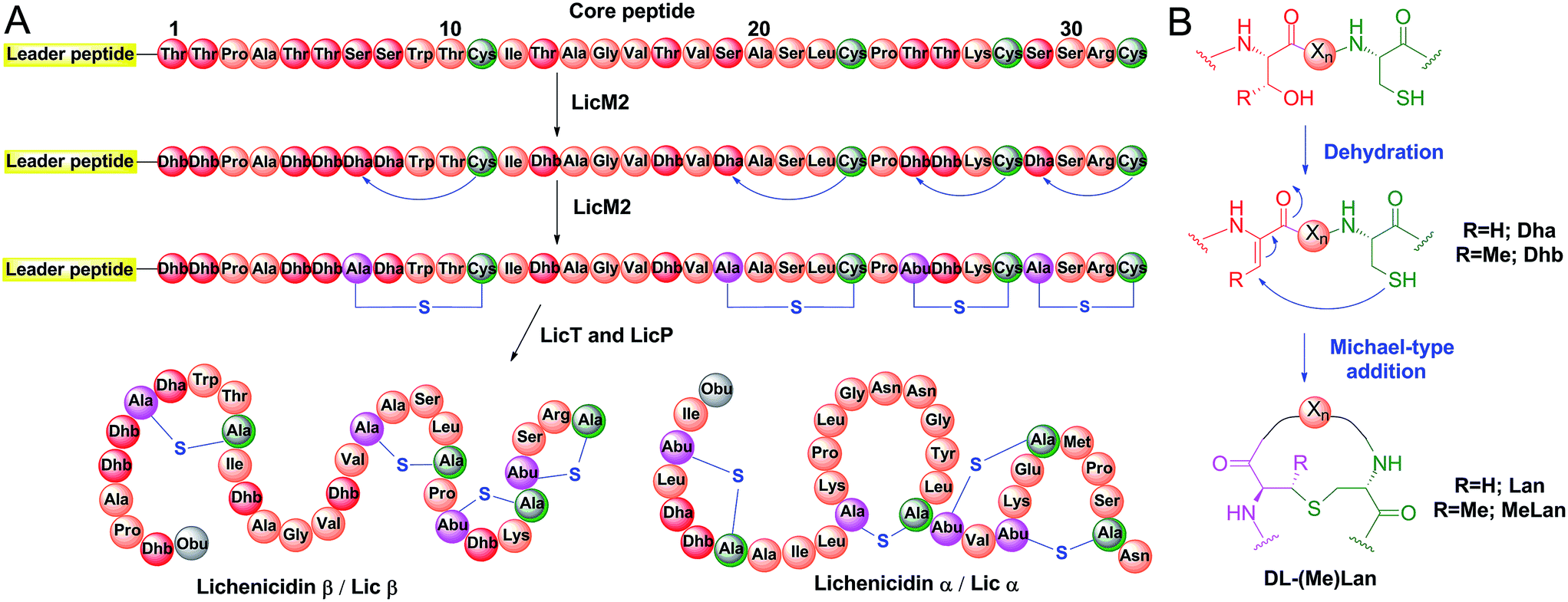
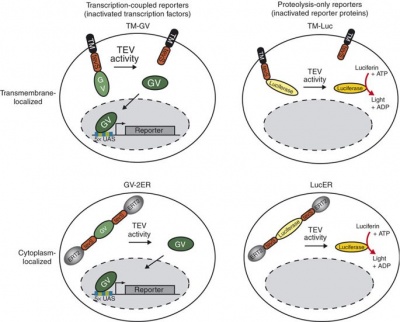
Applications of the class II lanthipeptide protease LicP for sequence-specific, traceless peptide bond cleavage - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02329G

Going native: Complete removal of protein purification affinity tags by simple modification of existing tags and proteases - ScienceDirect

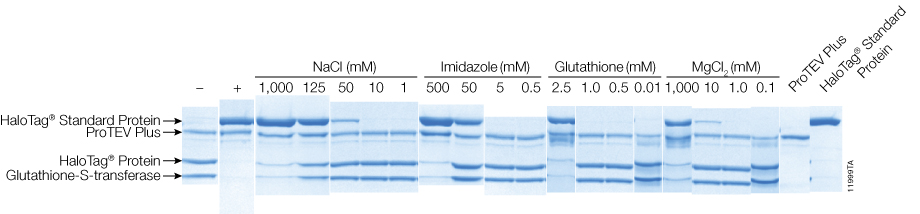
Phosphorylation regulates proteolytic efficiency of TEV protease detected by a 5(6)-carboxyfluorescein-pyrene based fluorescent sensor - ScienceDirect

TEV cleavage of Flag-tag. The TEV protease (brown) can be applied to... | Download Scientific Diagram
Recombinant Production of the Amino Terminal Cytoplasmic Region of Dengue Virus Non-Structural Protein 4A for Structural Studies | PLOS ONE